If you ever have the opportunity to visit a galvanizing plant and watch firsthand the hot-dip galvanizing process, you would gain a whole new appreciation for the “silver-looking poles”. It’s a fascinating process with so much behind the scenes chemistry going on.
To bring you up to speed, hot-dip galvanizing is the process of coating fabricated steel by immersing it in to a bath of molten zinc that metallurgically bonds the zinc to the steel. This practice has been around for over 150 years, and provides maintenance-free corrosion protection for decades
Quick Breakdown:
When the steel enters the kettle at 840 to 850 degrees Fahrenheit, zinc-iron alloy layers start to form. This portion normally represents about 50 to 70 percent of the total coating thickness, with the zinc being the top layer. Directly coming out of the kettle, all steel is extremely bright, despite the coating appearance.

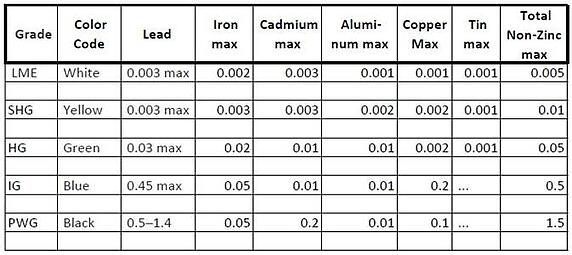
5 grades of zinc for continuous hot-dip galvanizing:
1. London Metal Exchange (LME) Grade- contains a minimum of 99.995% zinc
2. Special High Grade (SHG)- high purity of zinc containing a minimum of 99.990% of zinc
3. High Grade (HG)- contains a minimum of 99.95% zinc
4. Intermediate Grade (IG)- contains a minimum of 99.5%zinc
5. Prime Western Grade (PWG)- contains 0.5 to 1.4% lead, and a minimum of 98.5% zinc
There are distinctive looks and markings on the finished galvanized product depending on the grade used. For example, PWG lends itself to a more spangled look, whereas SHG has a more continuous bright coating.
When steel is delivered on-site the first thing that is noticed is the coating appearance. Upon further inspection, if discolored or lumpy areas are noticed, the most common concern is if it’s detrimental to the life span of the coating. But in many circumstances, the look tends to be more serious than the actual effects.
8 Most On-Site Concerns:
1. Bare Spots- Smaller flaws have little effect on the service life of the coating, and can be somewhat self-healing. Some spots may require repair using such methods indicated by ASTM A 780). But, uncoated, unrepairable spots can be grounds for rejection. Some causes of bare spots can be because of inadequate surface preparation, welding slag, rolling defects, sand embedded in castings or oxidized steel.
2. General Roughness- This is usually due to excessive growth or unevenness of the alloy layers, which can be attributed to the steel’s chemical composition or original surface condition. Heavy coatings are usually rougher than lighter coatings because irregularity of alloy layers tends to increase with thickness. In most cases, a rough coating does not negatively affect the lifespan, as long as adhesion is good. But, there are always exceptions to the rules. For particular pieces where one surface mates with another, rough coatings can be detrimental.
3. Dross Protrusions- Dross is the zinc-iron alloy that settles to the bottom of the kettle. It produces surface protrusions when the dross layer becomes agitated from the dross inclusions. Dross protrusions tend to have little effect on the surface life since the corrosion rate is similar to zinc. However, extensive dross inclusions can be grounds for rejection because they tend to make the surface more susceptible to mechanical damage.
4. Lumpiness and Runs- A lumpy coating results when the withdrawal is too fast or when the bath temperature is too low, not allowing molten zinc to drain back into the bath. Delayed drainage from bolt holes, folds, seams or other pockets where zinc collects is a consequence of the design. When products come in direct contact with others while being withdrawn from the kettle can also cause a lumpy coating appearance. Although it’s not detrimental to the life span, some cases require a smooth finish.

5. Flux Inclusions- Flux inclusions occur when a layer of zinc-ammonium chloride floats on the top of the molten zinc. When the steel is submerged in the bath, the flux pushes to the side when the steel is pulled back out. Flux inclusions can be caused by several different scenarios, such as a stale kettle flux where it tends to adhere to the steel instead of clearly separating from the surface as the steel is dipped. If the underlying coating is sound, then flux deposits are not reasons for rejection.
6. Ash Inclusions- Similar to flux, ash may be picked up during the dipping of the steel. Zinc ash is the oxide film on the surface of the bath. Ash inclusions can occur when steel requires slow withdrawal from the bath, and has no effect on the service life. If improper skimming of the exit surface of the bath can lead to gross oxide lumps, and can reduce the effective thickness of the coating, which is not acceptable.
7. Matte Gray or Mottled Coating- Usually appears as a localized dull patch or wed-like area on a normal surface, and develops when there is a lack of free zinc layer on the coating surface during the cooling process. A matte gray coating is found mostly on steel with relatively high silicon or phosphorous content, since they are heavier sections that cool slower. Galvanizers generally don’t have prior knowledge of the steel’s chemical composition, and has no control over its occurrence.
8. Rust Stains- Surface rust stains are not cause for rejection if they are caused by seepage from joints and seams after galvanizing or steel being stored under or in contact with rusty steel. Rust stains like this are superficial and should not be confused with failure of the underlying coat.
Whenever a question arises on the advisability of galvanizing a certain weld material, fabrication or steel type, it is best to consult the galvanizer. Most of the issues can be addressed beforehand if all parties stay in contact throughout the process before the steel arrives at the galvanizing plant. Remember to keep these 8 concerns in mind next time you conduct a visual inspection to help avoid delaying projects.


